 
Step 4: Draw the structure by writing already available valence electrons in pairs around each participating atom in a linear structure.īelow is the image of the lewis structure of the SeO2 molecule.Ĭan there be any other Lewis structure of Selenium Dioxide? This will be studied in detail under the polarity subheading. By this fact, selenium is the central atom.įurthermore, this can be confirmed by comparing the electronegativity values of selenium and oxygen.Īn element with the lowest electronegativity value becomes the central atom because it has to make the maximum number of bonds. The atom present as a single entity within a molecule is generally considered the central atom. 
Step 3: Find the central atom in the selenium dioxide molecule. So, the total number of valence electrons required by one selenium dioxide molecule is 24.īy this rule, only six valence electrons are further needed by one selenium dioxide molecule. 
Step 2: Find out the number of valence electrons further required to complete the octet of all three participating atoms in selenium dioxide.Īs per the octet rule, each atom needs eight valence electrons to complete its outermost shell. Valence electrons for selenium are six and for oxygen is also 6.Īs we have two oxygen atoms within one selenium dioxide molecule, the total number of valence electrons already present within one selenium dioxide molecule is 18. Step 1: Find out valence electrons already present within one selenium dioxide molecule. The Lewis structure begins with determining the number of valence electrons already available with one selected molecule and how many are further needed to achieve a stable condition. Steps to Draw the Lewis structure of Selenium Dioxide 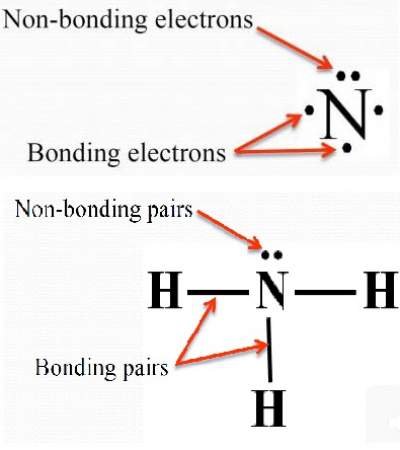
The need for such a structure is to get a preliminary idea of how atoms bond to produce a new chemical compound with new chemical properties altogether. In a typical structure, the atomic symbols of participating atoms are at the center, and valence electrons are in pairs around them. It uses dots to represent valence electrons and lines to show bonds. To sum it up! Lewis Structure of Selenium Dioxide (SeO2)Īlso called the Lewis dot structure, it is a pictorial representation of the behavior and arrangement of valence electrons within an atom. Let us study its lewis structure, geometry, and hybridization. There are various methods of preparing selenium dioxide, but the most preferred method is the dehydration of selenous acid. Immediate symptoms of selenium dioxide exposure to human beings include burning sensation, irritation to eyes, nausea, headache, etc. Even though selenium is beneficial for living organisms if consumed in smaller amounts but pertains to toxic effects such as deformed embryos and reproductive failure if exposed to larger quantities. Selenium dioxide reacts with water to produce selenic acid that initiates corrosion in most of the metals.īesides this, selenium dioxide reaches the soil and water through coal or oil combustion and weathering of rocks. This chemical compound is of great importance because of its corrosive nature for metals only when in contact with water. It is a unidimensional polymer chain having alternating selenium and oxygen atoms. The chemical formula of selenium dioxide is SeO2. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
